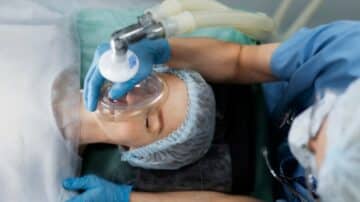
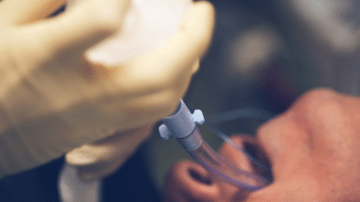
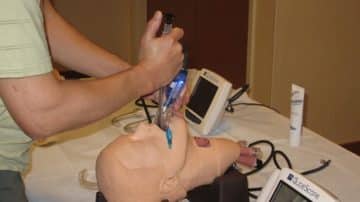
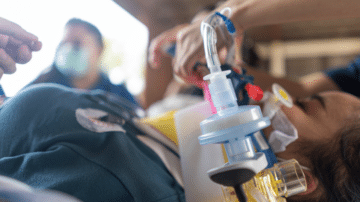
Endotracheal intubation can occur in up to 60% of critically ill patients. Though common, the procedure comes with significant risks. If multiple attempts are made to secure the airway, it may contribute to patient morbidity. Using the right laryngoscope can…
Read MoreProcedural sedation and analgesia (PSA) is a widespread practice that allows the patient to maintain oxygenation and independent airway control while being in a sedated or dissociative state. When should PSA be used and avoided? What are the various levels…
Read MoreNearly half of adult Americans are obese, putting them at higher risk for several health issues. Should they be admitted for treatment, this patient group is slightly more challenging to intubate than others (11% vs. 7%). The process requires clinicians’…
Read MoreWhich is better for critically ill patients—direct laryngoscopy (DL) of video laryngoscopy (VL)? This question has ignited an intense debate within academic, emergency medicine, and critical care communities as 13 to 20 million intubations are performed annually in the United…
Read MoreMore Tags – Breath Stacking
Mechanical ventilation (MV) is necessary for sustaining life among patients experiencing respiratory failure, cardiopulmonary arrest, severe neuromuscular disorders, upper airway obstruction, and those with unprotected airways. While mechanical ventilation can save lives, reduce mortality and lower healthcare costs, breath stacking…
Read MoreVideo laryngoscopy (VL) is a valid technique for difficult airway management in awake patients. But for patients with a history of difficult mask ventilation or intubation, awake fiberoptic intubation is widely considered the gold standard. It provides a clear visualization…
Read MoreMore Tags – ultrasound of spine
Longitudinal spine ultrasound with linear array probe Also known as a “spinal tap,” a lumbar puncture is used to diagnose life-threatening diseases, administer medications, and measure intracranial pressure. An estimated 360,000 of these procedures are performed annually in the U.S.…
Read MoreOver 400,000 Americans are intubated in emergency settings annually for various reasons—about 12.7% of them are unsuccessful on the first attempt. Failure to intubate the trachea on the first try occurs in up to 20% of cases. To make matters…
Read MoreMillions of people across the world experience thromboembolic diseases. It is a leading cause of death. Antiplatelet and anticoagulant agents used to treat these diseases are part of long-term anti-thrombogenic therapy plans. In fact, a wide range of medications have…
Read MoreCirrhosis refers to a medical condition where a patient’s liver is acutely scarred or damaged. It is irreversible and requires treatment to manage the condition and reduce the risk of liver failure. One in 400 adults suffer from liver cirrhosis…
Read MoreA lumbar puncture headache often develops after a patient undergoes a lumbar puncture or spinal tap procedure. Lumbar punctures with CSF analysis are often used to diagnose conditions such as meningitis, encephalitis, leukemia, autoimmune diseases, normal pressure hydrocephalus, benign intracranial…
Read MoreLiterature on spine ultrasound has grown over the last 22 years. Increased interest has led to updates in the technology used to evaluate spinal and paraspinal regions. Look at the available evidence with Hospital Procedures Consultants. Latest Updates in Spine…
Read MoreThere are several airway management techniques for securing airways in situations where the patient’s breathing is compromised. In cases where a difficult airway is anticipated, fiberoptic intubation is the method of choice since it has a high airway management success…
Read MoreIf patients’ lymphatic systems aren’t working efficiently, they can experience water retention (edema) in various parts of their bodies. Edema commonly occurs when patients have an infection, kidney disease, venous insufficiency, heart failure, or cirrhosis of the liver. This abnormal…
Read MorePleural effusions occur when excessive fluid such as blood or pus accumulates between the visceral pleura and parietal pleura of the pleural cavity. Evaluating patients for pleural effusions involves certain challenges. However, prompt diagnosis and treatment are essential to reduce…
Read MoreCategories
- ACLS (1)
- Arterial line (33)
- Cardiovascular diseases (77)
- Central line (55)
- Chest Tube (39)
- Dermatology (4)
- Emergency Procedures (138)
- Endocrinology (6)
- Endotracheal Intubation (36)
- Events (24)
- FAST Exam (12)
- Featured (112)
- Featured Procedure (42)
- Gastrointestinal diseases (32)
- Ginecology (3)
- Glidescope Intubation (21)
- Hematology (33)
- Hospital Procedures (85)
- Infections (32)
- Intraosseous line (8)
- King Tube (27)
- Laryngeal Mask Airway (18)
- Lumbar Puncture (36)
- Mechanical Ventilation (34)
- Medical General (95)
- medical procedures (258)
- Needle Decompression (6)
- Nephrology (11)
- Neurological diseases (12)
- Oncology (4)
- Paracentesis (32)
- Pericardiocentesis (3)
- Procedural Sedation (19)
- Respiratory diseases (85)
- RUSH Exam (8)
- Thoracentesis (37)
- Traumatology (24)
- Travel (27)
- Ultrasound-Guided Peripheral IV (13)